In September 2022, EPN launched the “Smart Leveraged Church Supply Chain” project, running until April 2025, to address critical challenges within the supply chains of health commodities in church-based health systems in Africa. The project aimed to overcome issues such as the lack of standardized tools to assess the maturity of Drug Supply Organizations (DSOs), the absence of a clear roadmap to improve performance among lower-level DSOs, and weak coordination across the supply chain. It also sought to address the limited progress in pooled procurement, workforce shortages in supply chain roles, and the lack of structured training for central-level staff. Whether it’s ensuring the right medication reaches a patient in a remote community or managing the inventory of a large hospital, the need for an efficient supply chain is clear.
In addressing the identified gaps, one of the objectives was to develop a mechanism to help DSOs assess and evaluate their supply chain maturity so as to be able to pinpoint ways of improving their delivery of health commodities.

Collaborative effort: Through the EPN TWG
To facilitate the development of this mechanism, EPN established a Technical Working Group (TWG) -Supply Chain and Quality Assurance (TWG) to review and gather insights from established frameworks and best practices. The TWG was composed of experienced professionals and experts drawn from EPN member organizations, ensuring the application of practical expertise and contextual relevance from their work.
To create a robust tool, the TWG chose the USAID NSCA 2.0 (United States Agency for International Development (USAID) National Supply Chain Assessment) NSCA 2.0 tool as the foundation for adaptation. The USAID NSCA 2.0 tool helps countries evaluate their existing supply chain systems, identify gaps, and determine opportunities for improvement to ensure that essential medicines, vaccines, and other critical health products reach the people who need them, especially in low- and middle-income countries. Through an iterative refinement, contextualization and incorporating feedback from various stakeholders, the TWG successfully adapted the NSCA 2.0 framework into the EPN DSO Maturity Assessment Tool.
A maturity assessment model is a tool for identifying the stage at which an organization, system or initiative has reached a scale of continuous improvement towards a desired end goal.
The EPN DSO MAT Tool?
The DSO Maturity Assessment Tool (MAT) is designed to help pharmaceutical organizations assess the maturity and effectiveness of their supply chain systems. It helps pinpoint both strengths and weaknesses within these key areas, offering actionable insights that can guide organizations toward improvement. The MAT tool allows organizations to:
- Evaluate their current supply chain systems.
- Identify areas for improvement.
- Develop strategies for enhancing supply chain performance.
Tailored to local contexts: One of the standout features of the tool is its flexibility. Developed with input from global supply chain experts and its extensive refinements, the final product is a well-rounded and robust solution, adaptable to local contexts and settings, making it suitable for use by pharmaceutical organizations of all sizes, including public and private sector organizations.
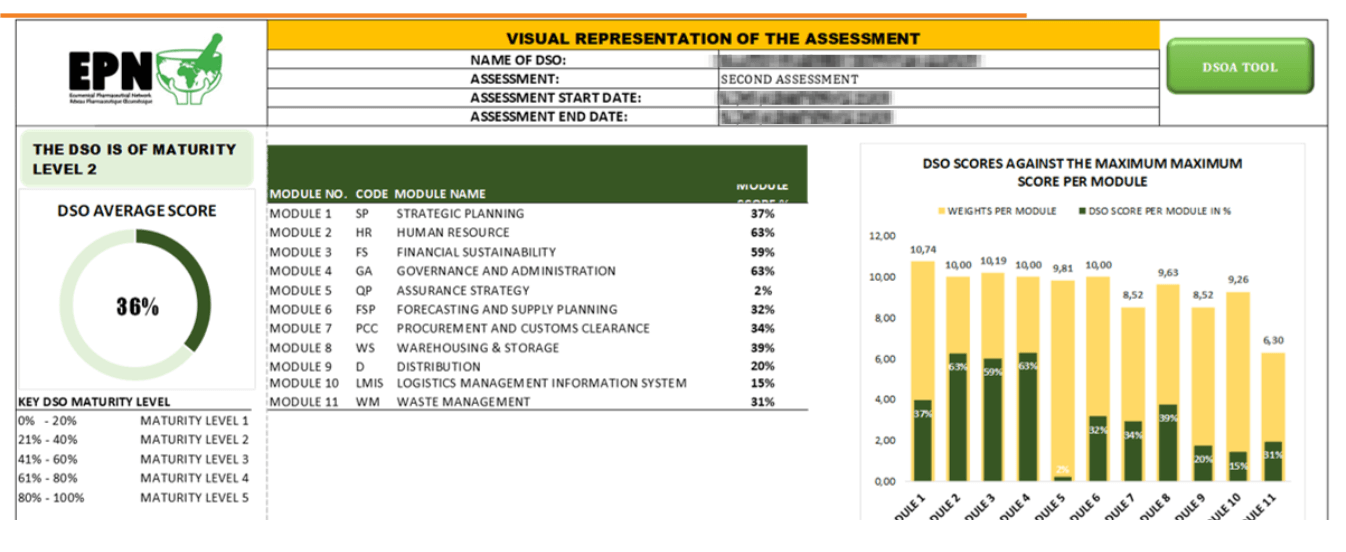
The tool defines 5 levels of maturity as defined below
| Maturity Level | Overall Score in Percent | Capacity Description at this level |
| 1 | 0 – 20 % | Characterized by unpredictable and reactive processes, with no defined strategies. |
| 2 | 21 – 40 % | Processes are only managed during transactions, which include basic automation. |
| 3 | 41 – 60 % | This stage involves reactive processes with clear strategies and systematic methods for managing supply functions. |
| 4 | 61 – 80 % | At this level, there is a clear demonstration of end-to-end visibility with measurable performance assessment using various tools. |
| 5 | 80 – 100 % | This represents the pinnacle of supply functionality, with optimized digital systems and a commitment to continuous improvements in all areas of supply performance. |
It comprises 11 essential modules, each addressing various aspects of a well-functioning supply chain management system.
- Strategic Planning (SP): Focuses on having a clear, actionable supply chain strategy, including budgets, monitoring plans and SOPs (Standard Operating Procedures).
- Human Resources: Assesses staff capacity and retention strategies, ensuring that key positions are filled with competent individuals and that ongoing training is in place.
- Financial Stability: Examines the organization’s financial health, including funding sources, with focus on reduced donor reliance and the management of unbudgeted costs.
- Governance and Administration: Reviews governance structures and the availability of essential manuals, policies, and guidelines for efficient decision-making.
- Quality and Pharmacovigilance: Ensures that medicines are of the highest quality and that safety monitoring systems (e.g., Pharmacovigilance) are in place including products Certificates of Analysis (CoAs) and conformance, and quality Assurance and related SOPs
- Forecasting and Supply Planning: Looks at inventory management, consumption data, and stock-taking procedures and visibility to prevent shortages or waste.
- Procurement and Customs Clearance: Focuses on the procurement process, supplier qualifications, internal controls and the smooth clearance of medicines at customs.
- Warehouse and Storage: Assesses storage practices, including temperature re controls and inventory checks, to guarantee product integrity.
- Distributions: Reviews distribution practices, policies and SOPs, such as route planning and the use of third-party logistics, to ensure timely deliveries.
- Logistics Management Information System (LMIS): Examines usage of paper or electronic systems for data tracking, data quality assessment and accurate reporting and availability of guiding policies and SOPs
- Waste Management: Establishes if proper systems are in place for pharmaceutical waste storage and disposal in line with regulations.
How can the MAT tool benefit pharmaceutical organizations?
- Improves decision-making: The tool facilitates organizational understanding of their supply chain strengths and weaknesses, in order to make informed decisions to boost efficiency and minimize risks.
- Enhances service delivery: It helps organizations streamline their processes, ensuring that quality medicines reach patients on time.
- Boosts cost savings: It enhances DSO e management, minimizes stockouts, and prevents overstocking, which ultimately saves costs.
- It is adaptable: The tool’s adaptability ensures that it’s useful in a variety of settings, from large hospitals in urban areas to smaller clinics in rural communities.
- It facilitates collaboration: The MAT Tool enhances involvement of stakeholders at every level, from senior management to field staff. This promotes a collaborative approach to supply chain management
How the MAT tool is utilized
The EPN DSO MAT Tool was initially developed with easily customizable platform for data entry, making it simple to track progress. Recognizing the need to expand its accessibility and adaptability, efforts are underway to adopt the tool into additional digital formats suited for diverse user environments and needs.
How has this tool been utilized?
Since its development, the tool has been disseminated and used to asses 3 DSOs in 3 countries that were participating in the project. Consequently, a number of gaps were identified, and a result, evidence-based roadmap was developed and implemented to strengthen DSOs supply chains, including a Monitoring and Evaluation tool to measure the progress.
The tool was also been used by seven (7) other DSOs to conduct self-assessments and determine their respective levels of supply chain maturity. A guideline is to be developed to take through the rest of the DSOs on how to utilize the tool.
Way forward
With the guideline for filling the tool developed, EPN is having the tool accessible to all member DSOs for their self-assessment. This will help to capacitate them to have stronger and resilient supply chains. Out of the MAT Tool results, there can also be learnings from the stronger DSOs by DSOs of lower levels, through benchmarking, to improve on their capacity.
Conclusion
The EPN DSO Maturity Assessment Tool represents a significant leap forward in strengthening supply chain systems for pharmaceutical organizations. The tool empowers organizations to identify weaknesses, optimize processes, and enhance service delivery. Its adaptability makes it a valuable resource across different contexts. EPN believes that as more DSOs adopt the tool, it fosters greater collaboration, drives cost savings, and ultimately ensures that essential health commodities reach those who need them most. Looking ahead, the EPN MAT Tool promises to be an essential catalyst in transforming supply chain management.
